
Iron is the most abundant element (by mass) making up Earth, while oxygen is the most common element in the Earth's crust. 
The following is a list of the 118 identified chemical elements. MS Excel(97) MS Excel(2007) PDF CSV Zip of all formats Original Page: click here. This contains list of 118 identified chemical elements. Submitted by editor on 7 March, 2014 - 14:47. There are 80 elements that have at least one stable isotope and 38 that have exclusively radionuclides, which decay over time into other elements. Download Excel File List of List of Chemical Elements. 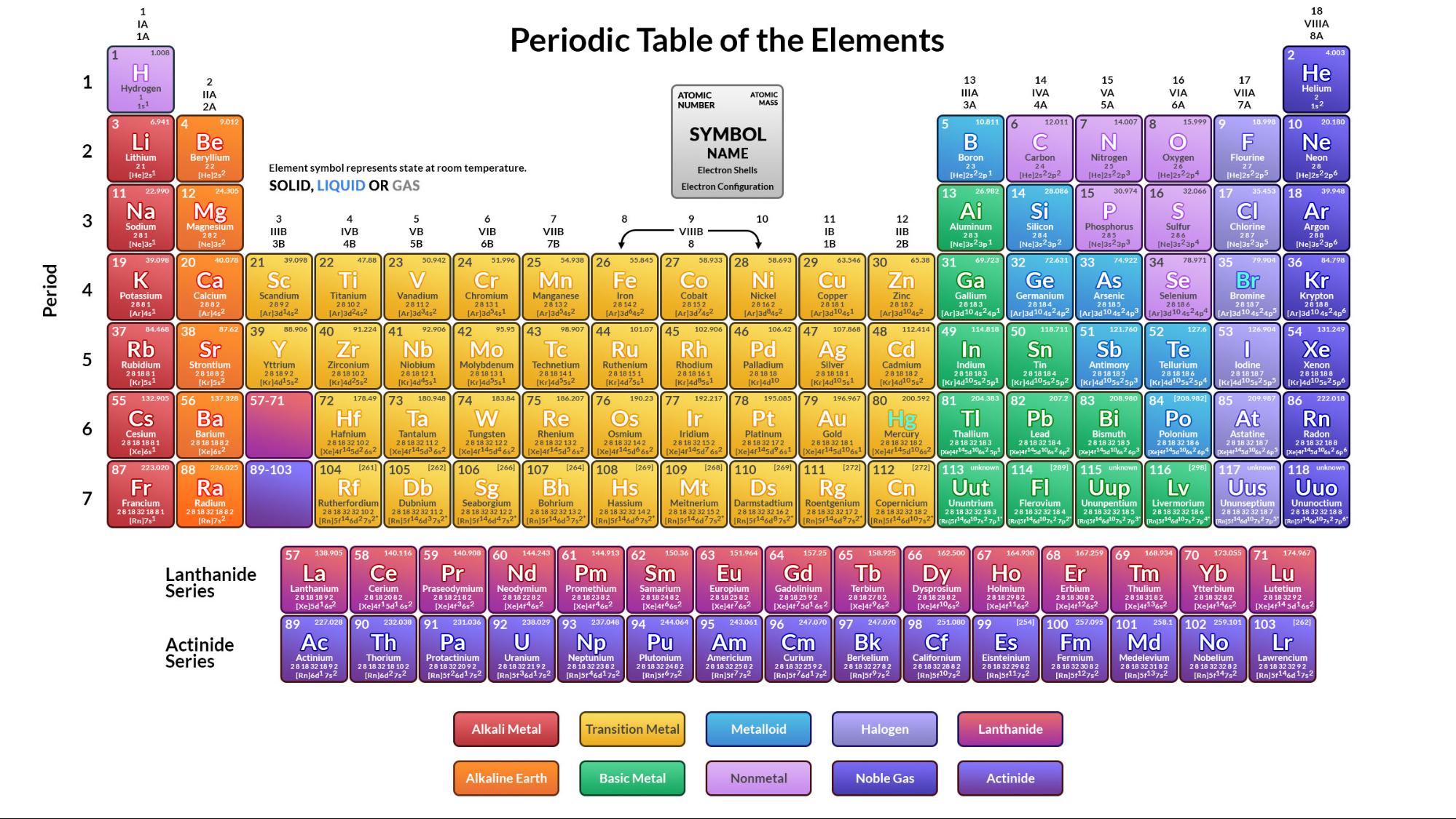
118 elements are identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There it stayed until 1940, when synthesis of the transuranium elements began.A chemical element is a species of atoms having the same number of protons in their atomic nuclei (that is, the same atomic number, or Z). The list quickly expanded to 92, ending at uranium (atomic number 92). By the time Russian scientist Dmitri Ivanovich Mendeleev (1834–1907) organized his periodic table in 1869, he had about 60 elements to reckon with. Berzelius published a table of 24 elements, including their atomic weights, most of which are close to the values used today.īy the year 1800 only about 25 true elements were known, but progress was relatively rapid throughout the nineteenth century. For example, writing two Hs and one O together as H 2O would mean that the particles (molecules) of water consist of two hydrogen atoms and one oxygen atom, bonded together. These symbols could be put easily together to show how the elements combine into compounds. Berzelius (1779–1848) was the first person to employ the modern method of classification: a one- or two-letter symbol for each element. 
Even though some of Lavoisier's "ele ments" later turned out to be compounds (combinations of actual elements), his list set the stage for the adoption of standard names and symbols for the various elements. In 1789, French chemist Antoine Lavoisier (1743–94) was able to publish a list of chemical elements that met Boyle's definition. 1577–c.1644) tried to explain everything in terms of just two elements: air and water.Įventually, English chemist Robert Boyle (1627–1691) revived Aristotle's definition and refined it. 1493–1541), also known as Paracelsus, proposed that everything was made of three "principles:" salt, mercury, and sulfur. For example, the Swiss physician and alchemist Theophrastus Bombastus von Hohenheim (c. Several other theories were generated throughout the years, most of which have been dispelled. C.), a student of Plato's, proposed that an element is "one of those simple bodies into which other bodies can be decomposed and which itself is not capable of being divided into others." Except for nuclear fission and other nuclear reactions discovered more than 2,000 years later, by which the atoms of an element can be decomposed into smaller parts, this definition remains accurate. C.) referred to these four "roots" as stoicheia elements. For instance, all of the elements in Group 1A are relatively soft metals, react violently with water, and form 1+ charges all of the elements in Group 8A are unreactive, monatomic gases at. C.) proposed that there are four basic "roots" of all materials: earth, air, fire, and water. In a periodic table arranged in order of increasing atomic number, elements having similar chemical properties naturally line up in the same column (group). The concept of elements-i.e., the theory that there are a limited number of fundamental pure substances out of which all other substances are made-goes back to the ancient Greeks.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
